Form and Function
Babesia canis is a cunning and well adapted parasite that has evolved in one of the most intelligent ways possible. This includes its relationship with the tick vector as well as the vertebrate host, primarily the dog. These relationships have aided in the movement, or transportation of this extremely well adapted species. B. canis has also formed special adaptations of structure that are unique to the many stages of its life cycle.
Specialized Structures
B. canis has
developed many specialized structures that fit certain functions.
While B. canis is within the tick vector in sporozoite form, it will
undergo further development and turn into a gametocyte. These
gametocytes have elongated bodies that have arrowhead shaped rays
characterize B. canis gametocytes, and are often called the
“raybodies”. These rays aid in the fusing into tick lumen, or
intestine, within the digestive tract. Once fused, the gametocyte
will develop into a zygote which has an arrow-shaped organelle which
releases enzymes that allow the zygote lyse through the digestive
tract (Mehlhorn and Schein. 1984).
Motile Stages for Transmission
B. canis has
two motile stages within its
life cycle. During true transmission
from vector to host, B. canis must be in sporozoite form. All other
parts of B. canis’s life cycle will deteriorate during transmission.
However, when B. canis is within the tick vector it has many
different stages that are mobile, each with a specific purpose.
These include ookinets, and sporokinetes. Ookinetes, products of
meiosis of the zygotic stage, are used to invade other tissues
beside the digestive tract of the tick. Ookinetes, in order to
undergo further development into sporozoites within the salivary
gland, must first become sporokinetes (Melhorn and Schein 1984,
Schein et al. 1980).
Adaptation for Lower impact in Host and Vector
B. canis is a
very well adapted organism. In fact, B. canis has evolved to have
the lowest negative impact on both vector and host bodies. It seems
that B. canis understands that if it were to heavily parasitize
either the host or the vector, that it as a population would suffer
greatly. Thus B. canis strives to stay at a low quantity within the
host and vector. Though staying at a low quantity a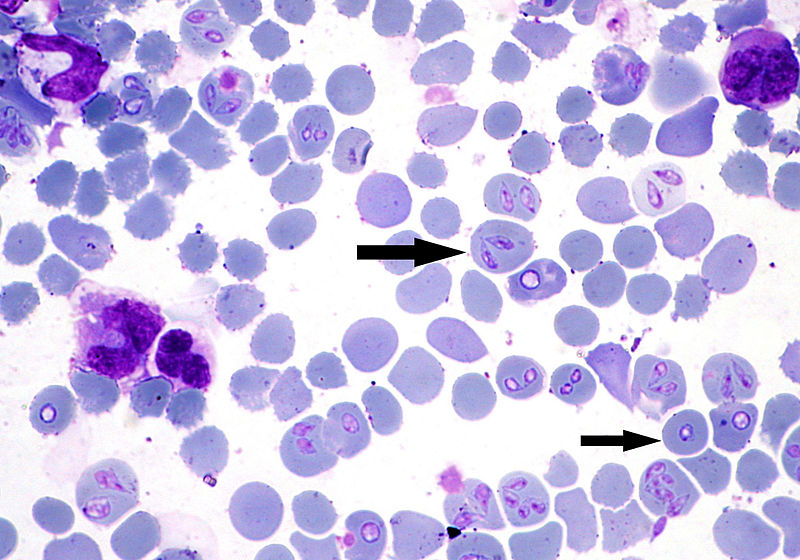 nd still
surviving within a host who’s immune system does not want to it to
be there is difficult. The host receives no benefit by having B.
canis within itself. This is important as the vector and host is key
to the transmission and thus the survival of B. canis within a given
ecosystem (Chauvin et al. 2009).
nd still
surviving within a host who’s immune system does not want to it to
be there is difficult. The host receives no benefit by having B.
canis within itself. This is important as the vector and host is key
to the transmission and thus the survival of B. canis within a given
ecosystem (Chauvin et al. 2009).
Transovarial and Transstadial Transmission
Two huge
adaptations for B. canis are Transovarial and Transstadial
Transmission. Transovarial Transmission occurs while B. canis is
within the tick in ookinetes form, in which the ookinetes infect the
eggs of a female tick. This allows B. canis to live and be spread to
hosts throughout the entire lifecycle of the tick, from conception
to death. This combined with the Transstadial transmission is a huge
advantage for B. canis. Transstadial Transmission occurs if
transovarial transmission does not occur. Transstadial, like
transovarial transmission, involve B. canis being present at every
stage of the tick’s lifecycle. Thus allowing B. canis to be present
during the reproduction stage of the tick’s lifecycle (allows
transovarial transmission to take place from this time on in
females)(Young and Morzaria 1986, Joyner et al. 1963).
To learn more about other oraganism that feed off of the
erythrocytes of other mammals, check out these other pages ,
Pediculus humanus,
Anopheles earlei, and
Ixodes scapularis.
Immune Response of Host
An immune
response in the canine host is caused by the injection of B. canis
sporozoites via tick blood meal. This response is intensified as the
sporozoites and the later merozoites divide through binary fission.
Though some dog breeds are more resistant then others. It was found
that the dog bread has a correlation to the resistance of B. canis.
B eagles, Fox Terriers, Dachshunds, and Mongrels are more resistant
to B. canis. Where as Spaniels, Griffons, Yorkshires Terrier, and
Doberman are less resistant. Though, once infected, if the dog is
treated and is able to rid itself of B. canis, the dog is able to
halt all forms of later infection (sterilizing immunity) (Martinod
et al. 1986, Brandao et al. 2003).
eagles, Fox Terriers, Dachshunds, and Mongrels are more resistant
to B. canis. Where as Spaniels, Griffons, Yorkshires Terrier, and
Doberman are less resistant. Though, once infected, if the dog is
treated and is able to rid itself of B. canis, the dog is able to
halt all forms of later infection (sterilizing immunity) (Martinod
et al. 1986, Brandao et al. 2003).
The canine host was once able to combat B. canis naturally
with the use of Bc28 proteins, however Bc28 is a very polymorphic
antigen, and B. canis has evolved since to become resistant to it.
Though further research has taken place and researchers have found
that a merozoite surface antigen (found in those dog breads that are
semi resistant to B. canis) has been found to prevent merozoites
from being able to lyse erythrocytes of the host, thus preventing
any further spread of B. canis. This will hopefully prove to be a
vaccine against B. canis in the near future (Carcy et al. 2006).
Immune response of Vector
The tick
vector is highly involved in the B. canis lifecycle. That being
said, the Tick does not benefit by having a parasite such as B.
canis within it. To counteract the B. canis, the tick has developed
multiple methods to counter, and destroy B. canis cells. The two
methods that are known to date are Longicine, and Longipain (Tsuji
et al. 2007, Tsuji et al. 2008).
Longicine is an antimicrobial peptide that is secreted into the tick digestive system where it adheres the membranes of merozoites (not common in the life cycle of B. canis while within the tick) and inhibits the proliferation of the parasite and kills it (Tsuji et al. 2007).
Longipain is rather similar to Longicine, though it is part of a network of enzymes. Longipain like Longicine will attack merozoites while within the tick but can be found in the mid-gut of the tick, and may adhere to the lumen of the digestive tract. Longipain does this by attacking B. canis in vitro by adhering to the membrane of B. canis (Tsuji et al. 2008).